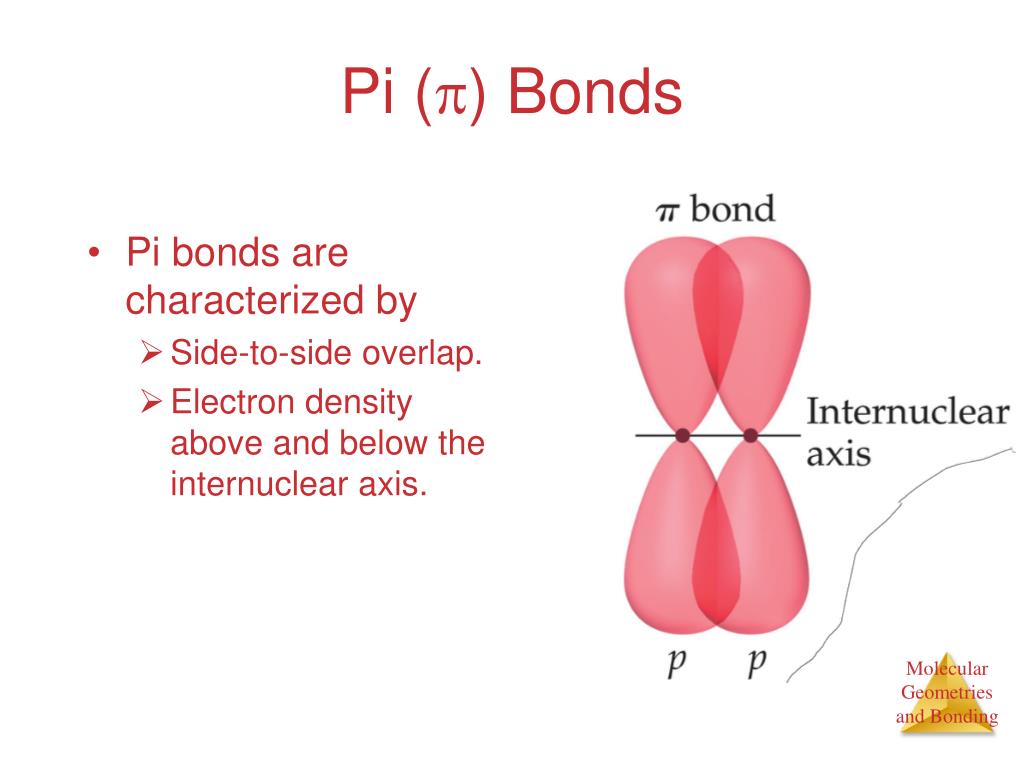
  Free rotation of atoms around pi bonds is not possible because it involves breaking the pi bonds. There can be free rotation of atoms around the sigma bonds. In other words, a single bond cannot be a pi bond. Thus, a pi bond is always present in molecules with multiple bonds, i.e., double or triple bonds. 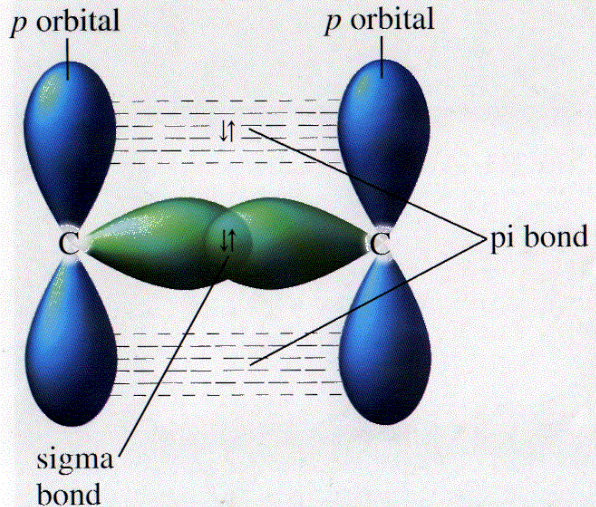
The reason is that the atoms constituting a single bond prefer to form a strong sigma bond rather than a weak pi bond. electrons, number of electrons, sigma bonds and pi bonds, sigma-bonds, pi. For example, ethylene molecule contain 5 sigma bonding and 1 pi bonding. chemical bonding, chemistry of life, electrode potential, electrons in atoms. They are formed based on the orbitals of the bonding electrons between two atoms, i.e. The covalent bond formed by lateral overlap of atomic orbitals is called as pi bond. The reason is that the overlapping of atomic orbitals can take place to a greater extent during the formation of a sigma bond, whereas overlapping of orbitals occurs to a smaller extent during the formation of a pi bond.Ī pi bond between two atoms is formed only in addition to a sigma bond. A sigma bond is a single bond, a double bond is a sigma and pi bond, and a triple bond is a sigma bond and two pi bonds. 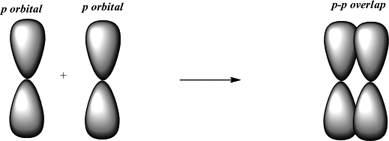
The orbital overlap takes place in such a way that their axes are parallel to each other but perpendicular to the internuclear axis.Ī sigma bond is stronger than a pi bond. They are made from hybridized orbitals.Pi bonds are the SECOND and THIRD bonds to be made. This type of covalent bond is formed by the lateral or sideways overlap of the atomic orbitals. Sigma bonds are the FIRST bonds to be made between two atoms. The atomic orbitals overlap along the inter-nuclear axis and involve end-to-end or head-on overlap. This type of covalent bond is formed by the axial overlapping of half-filled atomic orbitals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
